WILLIAM CHILDRESS - SCHOOL OF RENEWABLE NATURAL RESOURCES
Project Summary
With respect to the cryopreservation of genetic resources of aquatic species, research activity is restricted to producing data for publication. Small-scale application can produce working material (samples) with low-to-medium throughput, typically without quality assessment. Commercial activities would produce products, potentially numbering in the millions, with high levels of quality management (QM). These are qualitatively different levels of activity, and research does not equal application, nor does it necessarily lead to it. Transition across these levels requires the establishment of new capabilities. Cryopreservation within aquatic species has predominantly remained arrested in the research phase for the past 70 years, but has begun the transition to small-scale use, primarily at stock center repositories developed to support specific research communities, for example, the Zebrafish International Resource Center (ZIRC, www.zebrafish.org) for biomedical research. The primary approach for this small-scale level is the central facility concept where samples or animals are transported to a specialized facility with expensive equipment and specifically trained personnel.
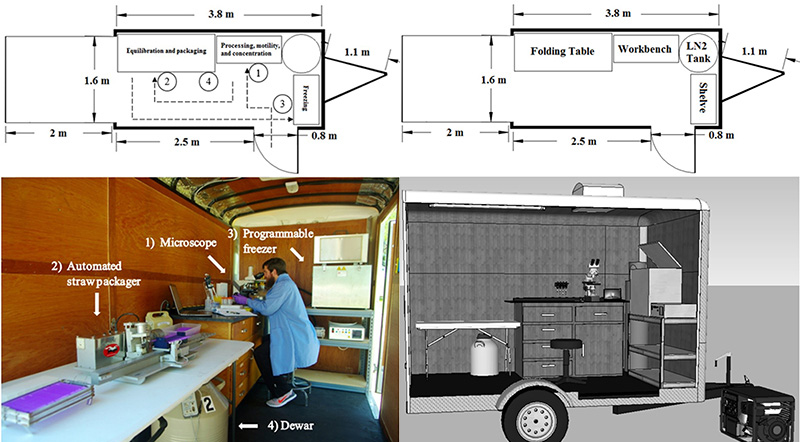
A second model for moving material into repositories would be a mobile laboratory concept. However, this is still at the research level with little analysis to expand it to small-scale application. This has constrained development of aquatic species repositories because it is often difficult to transfer animals or samples, and can result in loss of gamete quality yielding poor source material at the start of the cryopreservation process. More work is needed to evaluate the mobile laboratory concept to examine capabilities and requirements, and to bring it into the reality for routine application. Such work would include feasibility for scalable medium-to-high throughput, and would include biosecurity and QM components encompassing Quality Control (QC) and Quality Assurance (QA). This concept should include sufficient flexibility and adaptability to address a wide range of species, environments and host facilities.
For sperm cryopreservation, quality control can include repeated motility and cell concentration estimations, membrane integrity assessment, and disease screening. These activities help increase the repeatability and consistency of operations among laboratories and institutions. This is a key component for the development of germplasm repositories. Biosecurity programs for cryopreservation, especially for aquatic species, have to account for disease transmission, introduction of exotic species, and potential genetic consequences in the present and into the future when samples are used. Improper sample storage in the field or during transportation to and from working locations can increase the possibility of negative outcomes. Also, because most fish release sperm and eggs directly in the water to spawn, biosecurity should address potential effects to surrounding environments, such as the release of endemic or non-endemic pathogens
The overall goal of this study was to work with different user groups and cryopreserve sperm on-site at their facilities to evaluate the uses and challenges of a mobile laboratory with high-throughput and quality control capabilities comparable to those of a specialized centralized facility. The objectives were to demonstrate collection and cryopreservation of sperm of: 1) large-bodied freshwater Blue Catfish (Ictalurus furcatus) for aquaculture; 2) small-bodied freshwater Xiphophorus for biomedical and imperiled repository development, and 3) saltwater Red Snapper (Lutjanus campechanus) for wild fisheries research. Over the course of this project, the mobile laboratory traveled more than 4,000 km collecting germplasm from more than 650 male fishes. A total of 136 Blue Catfish were processed in 2015 and 2016 resulting in a total of 6,146 0.5-mL French straws. A total of 521 males from 11 different species in the genus Xiphophorus were processed over 4 d in 2015 resulting in a total of 488 0.25-mL French straws. And, a total of 17 Red Snapper males were processed during 2015 resulting in a total of 316 0.5-mL French straws. This is the first development of a mobile laboratory with high-throughput capability for aquatic species. User groups would no longer be limited to germplasm resources that can only be shipped as samples or transported as live animals to a central cryopreservation facility. Mobile laboratories create opportunities to collect higher quality germplasm, provide access to new species, and enable direct cooperation, including training, with a wide variety of user groups and applications.